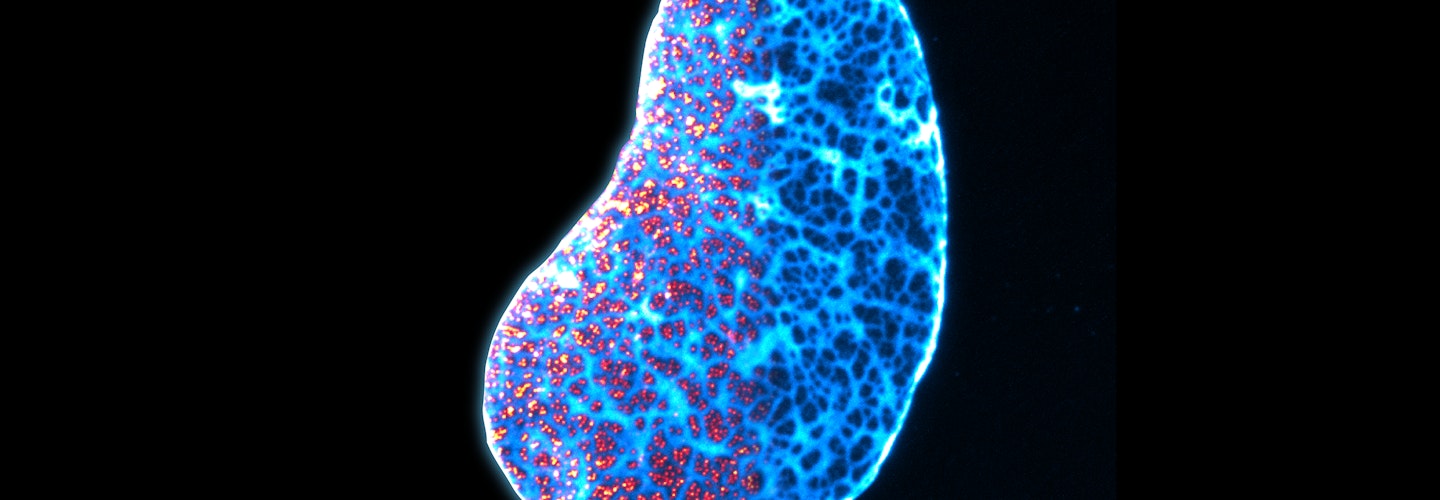
By Stephani Otte, Science Program Officer, Imaging, Chan Zuckerberg Initiative
Fifty years ago, when doctors saw patients with cancer, they had very limited information to make their diagnosis: inconclusive symptoms, maybe family history, and other indicators that could point toward lots of different conclusions. But thanks to the imaging technologies we have today—think MRIs and X-rays—doctors can look inside our bodies, identify where the tumor is located, and hopefully treat it before it's too late.
The problem is, we typically go through this process once a disease is progressing. A cell already became cancerous and started undergoing rapid division to form a tumor, and that tumor resulted in other symptoms that, finally, prompted a doctor’s visit.
But what if we could get a read on the system before the cancer — or any other disease — becomes symptomatic? What if we could view cells in high-resolution, watch them interact in real-time, and intervene in the pre-diagnosis stage instead?
Doing so would require imaging tools that go beyond our current capabilities. Some technologies give us a clear snapshot of the body, but they can’t show us what’s happening in real-time. Other tools can show us what’s happening moment-to-moment, but not at the resolution we need.
These limitations can have dire consequences. In a research setting, they can slow our progress toward new discoveries. In a clinical setting, they can mean the difference between a diagnosable disease and a mystery illness—a problem we can solve and one we can’t. For some patients, the stakes are life and death.
That reality should push us toward a new paradigm in biomedical imaging. Instead of accepting the tradeoffs between one technology and another, we must improve them and build new ones. Our objective should be to observe and measure human biology in real time, from entire organs to molecules, in high resolution.
Amazingly, much of what we need has already been built. Advanced microscopy is revealing the structure of living cells with breathtaking clarity; machine learning algorithms are making it easy to analyze the most massive biological datasets. To be sure, there are more technologies we need to invent. But at this point, the bigger challenge may be marrying them all together—and getting them into the hands of researchers and clinicians around the world.
That’s what my organization, the Chan Zuckerberg Initiative (CZI), aims to do. We recently announced that we’re creating a state-of-the-art advanced biomedical imaging institute in the San Francisco Bay Area. There, biologists, physicists, engineers, and data scientists will work together to build the next generation of bioimaging technologies. Over the next 10 years, we want to develop technology to comprehensively visualize processes in the human body, across scales and in real time.
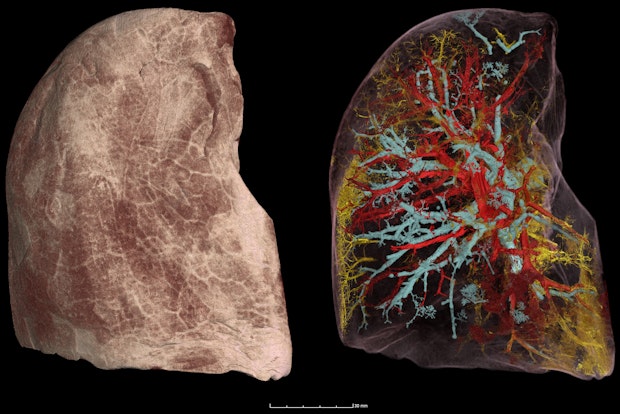
While that goal is undoubtedly ambitious, we know that rapid progress is possible. Take, for example, HiP-CT scans, which were invented during the pandemic to produce extremely high resolution images of human organs. When scientist Paul Tafforeau and his team used HiP-CT to scan the lungs of COVID-19 victims, they were able to map the virus’s effects at the cellular level—and dramatically advance our understanding of how it impacts the body.
What if we could view healthy and diseased tissue at an even higher level of resolution, down to the individual proteins within a cell?
It’s a question of enormous biological interest; proteins are responsible for all cellular function, and we don’t yet have the tools to measure their activity in real time. If we did, we could begin to answer any number of pressing biomedical questions—from how drugs affect brain cells that are undergoing neurodegeneration, to how viruses hijack our cellular machinery.
With that in mind, the imaging institute’s first major project will be to develop the technology to comprehensively map proteins and other macromolecules in cells, statically and dynamically—something that’s never been done before. This work will build on the progress of CZI’s existing support for visual proteomics.
In this project and others, the institute will also work to share its tools, methods, and data with the global biomedical community—because open science leads to more breakthroughs.

This is certainly what we’ve seen with a free, open-source tool called napari. This community-built platform helps researchers analyze and share the high-dimensional datasets from modern imaging devices. It’s also what we’ve seen with the napari hub that CZI is building, where imaging experts build and share software plug-ins with one another—like tools to measure SARS-CoV-2 antibodies.
When we prioritize accessibility to technology, we also advance equity. Historically, scientists in countries with relatively high investments in research and development—many of them in North America and Europe—have had far greater access to bioimaging tools than scientists elsewhere. This has skewed the global research agenda and slowed progress that would benefit all of us. But the next generation of technologies could help change that.

Guided by these open science principles, we will continue growing and learning from bioimaging communities around the globe. Through three-year grants, we're supporting scientists in Africa, Latin America, and Eastern and Southern Europe as they build capacity, regionally-coordinated infrastructure, and networks with other imaging experts. This resource-sharing approach can lower costs and boost health outcomes—where these scientists are based and across the entire region.
All of this will help advance our larger objective at CZI Science: to support science and technology that will make it possible to cure, prevent, or manage all diseases by the end of the century. It’s an ambitious goal. But the deeper and more accurately we can see into the human body, the clearer our path becomes.
___________________________________________________
Chan Zuckerberg Initiative is a 2022 Aspen Ideas: Health underwriter. The views and opinions of the author are their own and do not necessarily reflect those of the Aspen Institute.


